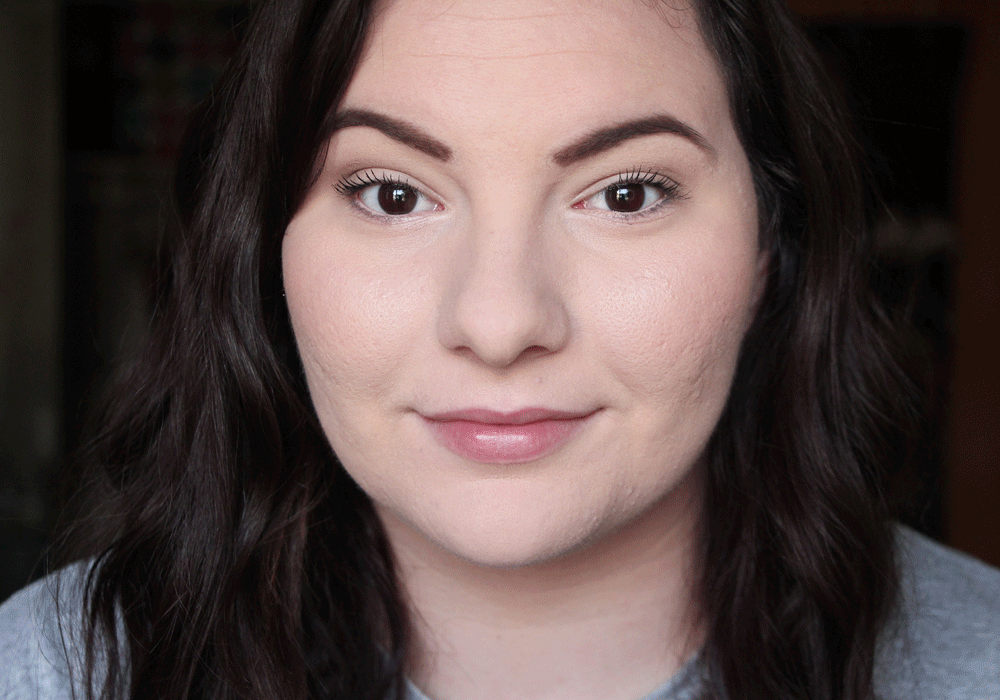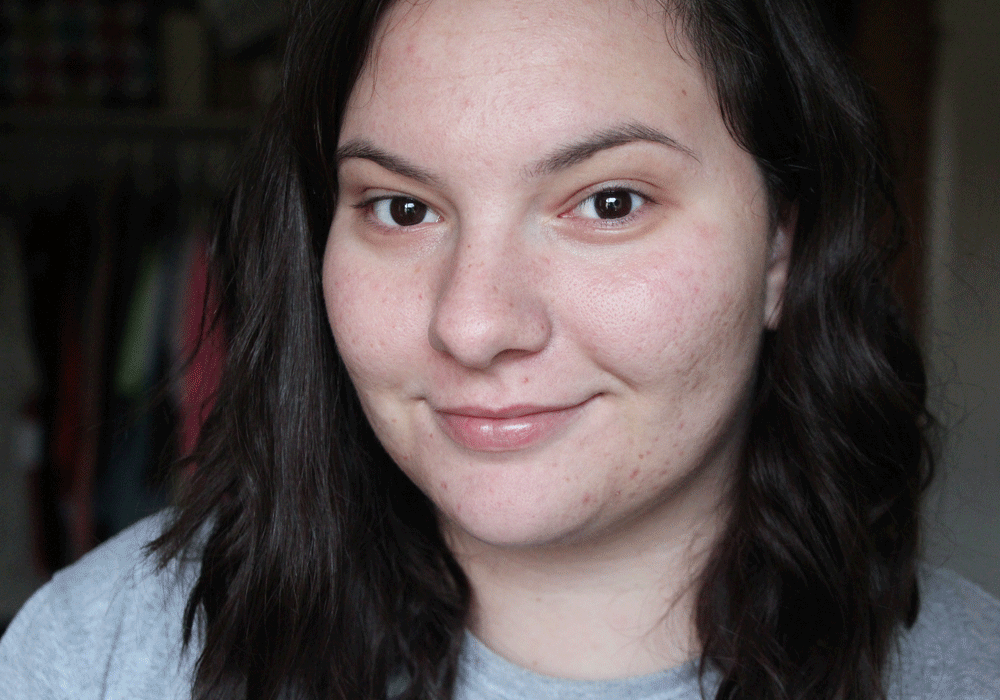Disclosure: This post was sponsored by Galderma Laboratories L.P. through their partnership with POPSUGAR. While I was compensated by POPSUGAR to write a post about Mirvaso, all opinions are my own.
As a beauty blogger, my skin is very important to me. I have struggled with skin issues since the moment I hit puberty, because I had a lot of acne. To this day, I still have occasional acne, but the scars are still left behind from my teenage years. Although I have never experienced rosacea, I can definitely relate to the confidence issues that having a skin condition can cause. Although it’s tempting to cake on makeup to disguise the problem (which I definitely am guilty of- it’s the reason I started getting into makeup, actually!), there is a way to conceal the redness without it. Mirvaso is the only FDA-approved treatment for redness caused by Rosacea. It provides same-day results that last up to 12 hours before the redness returns, making it ideal for days and nights out! They are launching a “Break Up With Your Makeup” campaign to raise awareness of the condition and new treatment and are hosting a contest to win a trip to the Emmy’s!
If you want to see more information about Mirvaso and the contest, please head on over to their site!: http://mirvaso.com/sweepstakes?v=1
Important Safety Information
Indication: Mirvaso® (brimonidine) topical gel, 0.33% is an alpha adrenergic agonist indicated for the topical treatment of persistent (nontransient) facial erythema of rosacea in adults 18 years of age or older. Adverse Events: In clinical trials, the most common adverse reactions (≥1%) included erythema, flushing, skin burning sensation and contact dermatitis. Warnings/Precautions: Mirvaso Gel should be used with caution in patients with depression, cerebral or coronary insufficiency, Raynaud’s phenomenon, orthostatic hypotension, thromboangiitis obliterans, scleroderma, or Sjögren’s syndrome. Alpha-2 adrenergic agents can lower blood pressure. Mirvaso Gel should be used with caution in patients with severe or unstable or uncontrolled cardiovascular disease. Serious adverse reactions following accidental ingestion of Mirvaso Gel by children have been reported. Keep Mirvaso Gel out of reach of children. Not for oral, ophthalmic, or intravaginal use. You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.